News
La Fe Health Research Institute studies the cardiotoxicity of anthracyclines in paediatric cancer using cardiac cells derived from the patients themselves.
The research team has generated induced pluripotent stem cells from samples from two paediatric patients with acute anthracycline-induced cardiotoxicity
The study provides information to develop more effective therapeutic strategies to reduce cardiotoxicity and improve treatment safety
Researchers from the Cardiac Regeneration and Transplantation and Translational Research in Cancer groups at the La Fe Health Research Institute (IIS La Fe) have managed to model anthracycline cardiotoxicity in oncopediatrics by studying the cardiomyocytes of two paediatric cancer patients who had heart problems due to anthracyclines.
Anthracyclines are a group of drugs used in the treatment of cancer. They are among the most common chemotherapeutic agents and one of their adverse side effects is cardiotoxicity, a significant clinical problem associated with patient survival.
In the case of childhood cancer, as the number of survivors increases, so does the number of people who may suffer cardiac damage as a result of anthracycline treatment. Therefore, there is a continuing need for new models to better understand the mechanisms of anthracycline-induced heart damage.
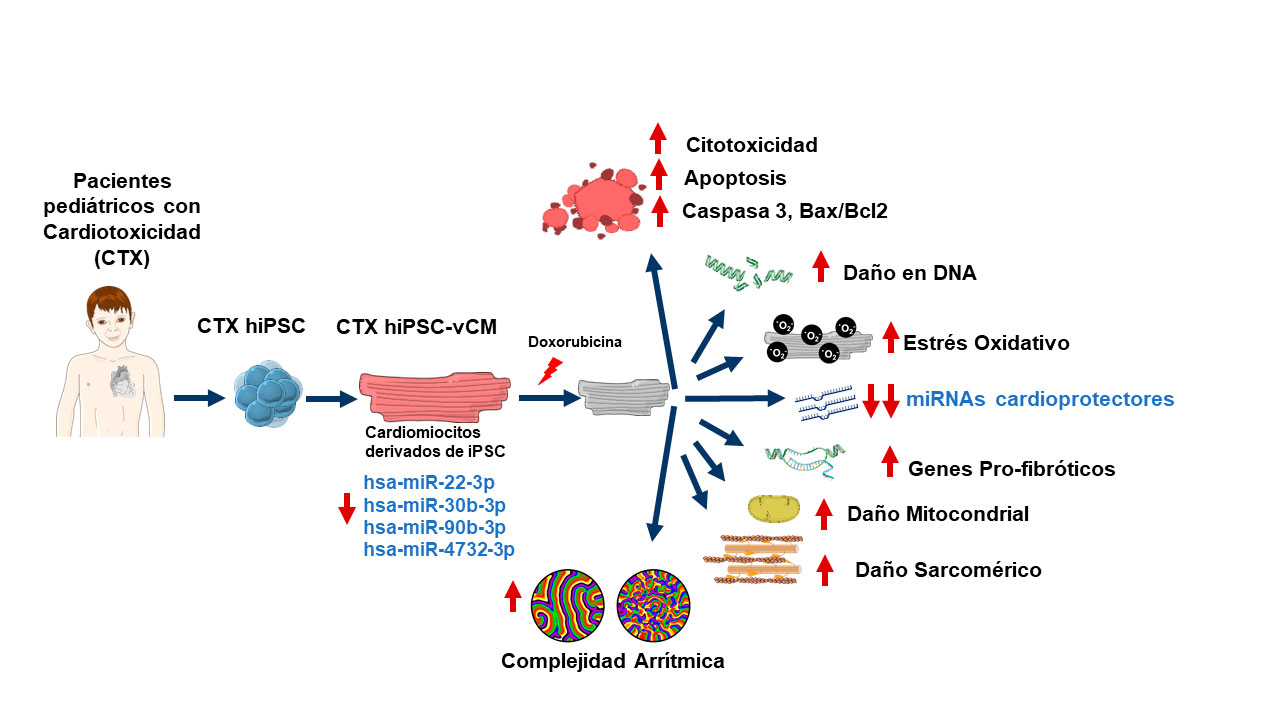
To this end, the research team at IIS La Fe has created induced pluripotent stem cells (iPSCs) from the blood of two paediatric cancer patients who had heart problems due to anthracyclines. iPSCs are a special type of cell that have been reprogrammed in the laboratory from adult cells, such as skin or blood cells, and have the ability to transform into almost any type of cell in the body. In this case, iPSCs have differentiated into ventricular cardiomyocytes, a specific type of muscle cell found in the ventricles of the heart.
Anthracycline-sensitive cardiomyocytes
Comparing these ventricular cardiomyocytes with cardiomyocytes from healthy subjects, the researchers found that cells from patients with cardiotoxicity were much more sensitive to damage caused by the anthracycline doxorubicin (DOX). These cells showed signs of programmed cell death, increased oxidative stress and damage to DNA and mitochondria. They also showed alterations in cell structure.
The study also observed that the expression of several miRNAs involved in structural integrity and inflammatory response was differentially affected by anthracyclines in cardiac muscle cells from patients with cardiotoxicity and this resulted in increased complexity of arrhythmias after anthracycline treatment.
This study provides valuable information on how the heart is affected by anthracycline treatment and highlights the importance of using patient-specific heart cells to study and understand these problems. By using these personalised heart models, researchers can look for ways to protect the heart from damage caused by these cancer drugs, ultimately improving care for paediatric cancer patients. However, more research is needed to fully understand all the mechanisms involved in this process.
The research involved the participation of the team of paediatric oncologists from the Hospital Sant Joan de Déu and researchers from the Centre for Regenerative Medicine in Barcelona.
Reinal I, Ontoria-Oviedo I, Selva M, Casini M, Peiró-Molina E, Fambuena-Santos C, Climent AM, Balaguer J, Cañete A, Mora J, Raya Á, Sepúlveda P. Modeling Cardiotoxicity in Pediatric Oncology Patients Using Patient-Specific iPSC-Derived Cardiomyocytes Reveals Downregulation of Cardioprotective microRNAs. Antioxidants (Basel). 2023 Jul 3;12(7):1378. doi: 10.3390/antiox12071378. PMID: 37507917; PMCID: PMC10376252.







